1st of September 2022
PRESS RELEASE
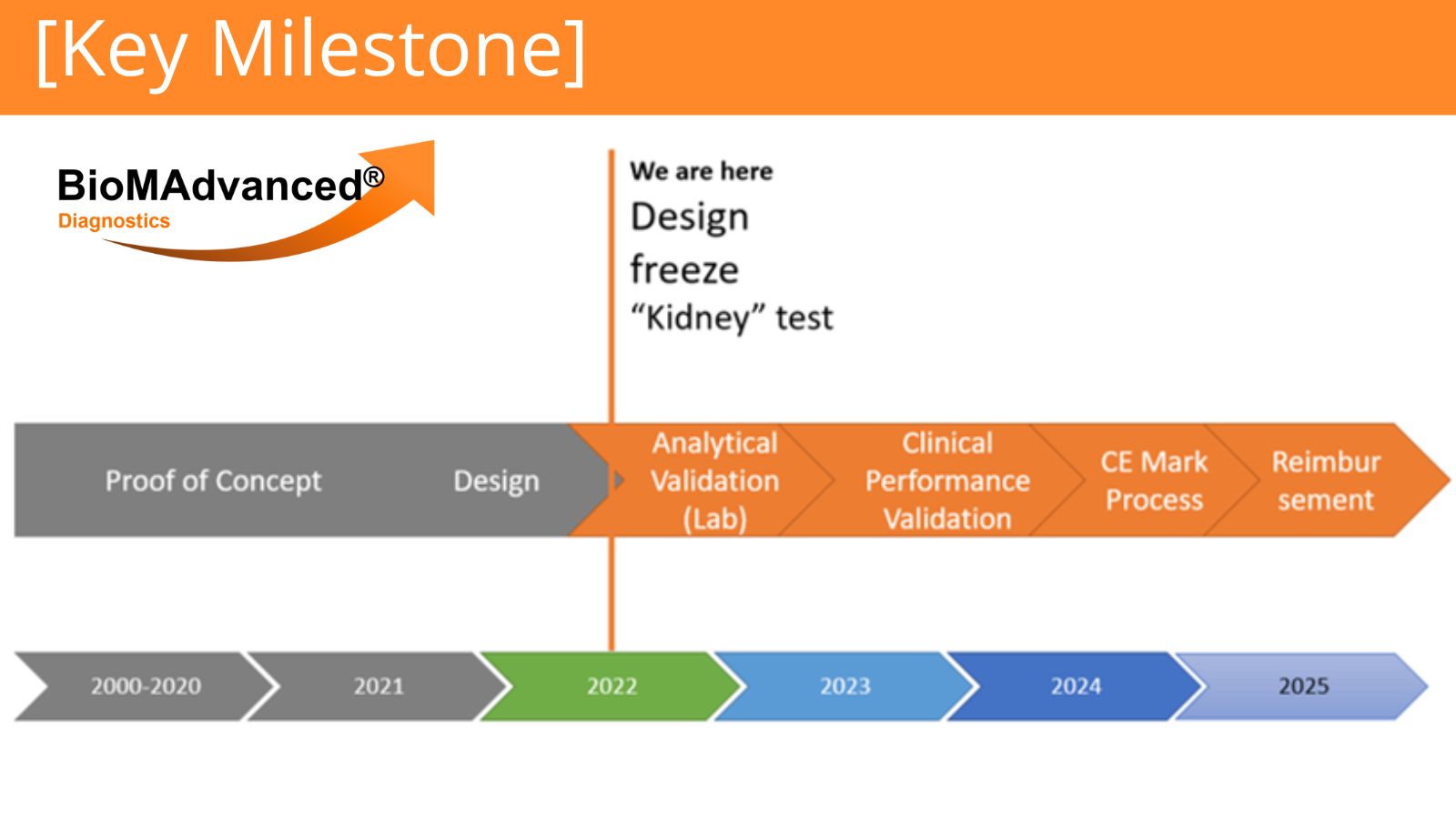
Major Milestone: BioMAdvanced Diagnostics is extremely proud to announce the Design Freeze of its IVD (*) test for Transplant kidney rejection!
This Milestone concludes the Design phase which took a translational research proof of concept and transformed it into a proper medical grade product.
Another source of pride is the fact that we carry on respecting the schedule set at the company inception. This design freeze also enables us to project the future steps with confidence. We therefore reiterate our projection to have this product CE marked by 2025.
This original phase is also extremely important as it serves as the foundation on which the other 2 products planned are going to be built. We estimate that the company will need approximately half the time to finalize the design of its IVD test for Transplant Lung rejection, which is already started.
Now, the analytical validation for the Kidney product is about to start, more on this in our next communication!
(*) IVD : In vitro diagnostics